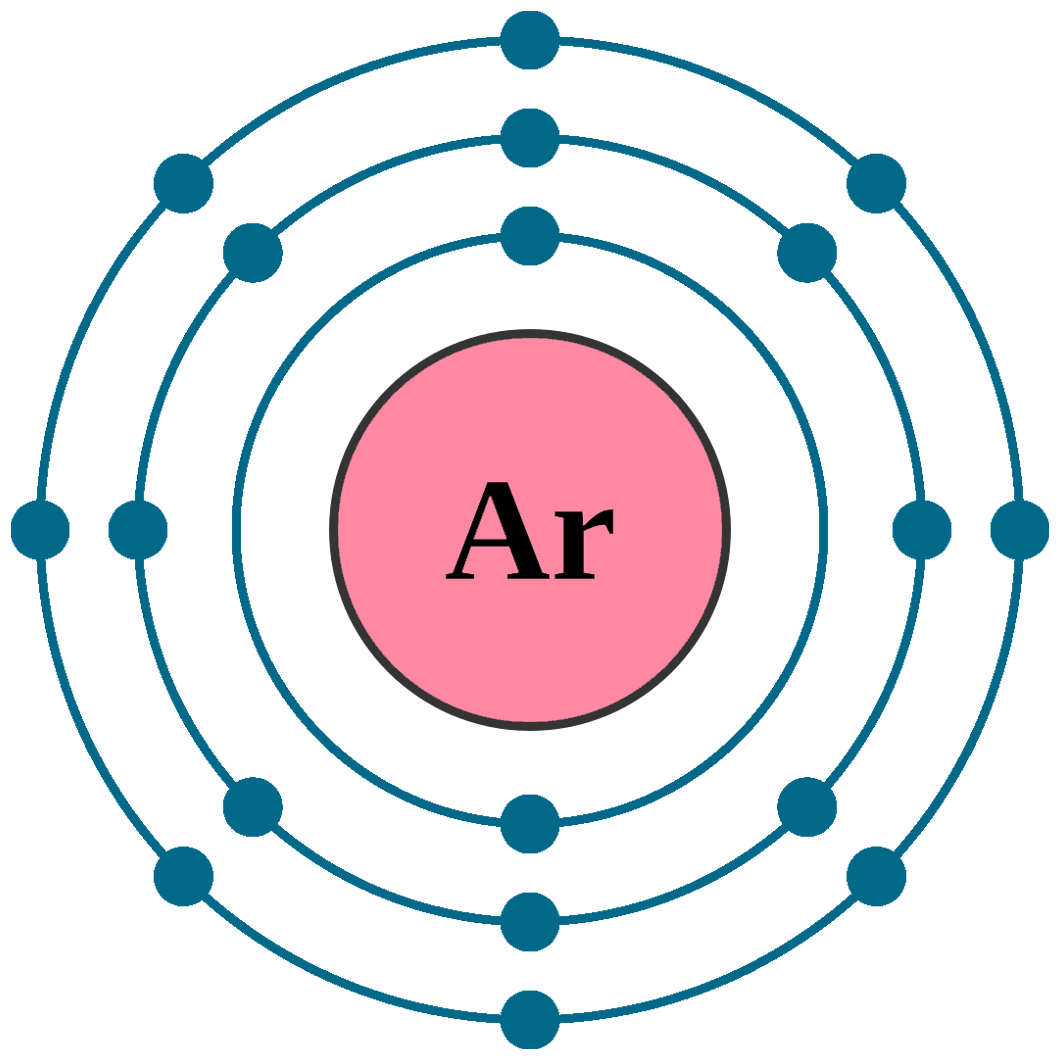
Argon is a fascinating element with a rich history and a variety of applications. It is a noble gas, meaning it is chemically inert and does not readily form compounds with other elements. One of the key aspects of argon that makes it unique is its Argon # of Neutrons. Understanding the number of neutrons in argon atoms provides insights into its atomic structure and properties.
Understanding Argon's Atomic Structure
Argon is the third element in Group 18 of the periodic table, following helium and neon. Its atomic number is 18, which means it has 18 protons in its nucleus. The most common isotope of argon is argon-40, which has 22 neutrons. This makes the total number of nucleons (protons + neutrons) in an argon-40 atom 40.
The number of neutrons in an atom can vary, leading to different isotopes of the same element. For argon, the most abundant isotopes are:
- Argon-36: 18 protons + 18 neutrons
- Argon-38: 18 protons + 20 neutrons
- Argon-40: 18 protons + 22 neutrons
Argon-40 is the most abundant isotope, making up about 99.6% of natural argon. The other isotopes, argon-36 and argon-38, are present in much smaller quantities.
The Significance of Argon # of Neutrons
The Argon # of Neutrons plays a crucial role in determining the element's properties and behavior. The number of neutrons affects the stability of the nucleus and the overall mass of the atom. For argon, the stability of its isotopes is a key factor in its applications.
Argon-40, with its 22 neutrons, is particularly stable and does not undergo radioactive decay. This stability makes argon-40 ideal for use in various industrial and scientific applications. For example, argon-40 is commonly used in welding and metal fabrication due to its inert nature, which prevents oxidation and contamination of the materials being worked on.
In contrast, argon-36 and argon-38 are less stable and can undergo radioactive decay, although their half-lives are extremely long. This means that while they are present in natural argon, their quantities remain relatively constant over time.
Applications of Argon
Argon's unique properties, including its Argon # of Neutrons, make it valuable in a wide range of applications. Some of the most common uses of argon include:
- Welding and Metal Fabrication: Argon is used as a shielding gas to prevent oxidation during welding processes. Its inert nature ensures that the weld remains pure and strong.
- Lighting: Argon is used in incandescent and fluorescent lighting. It helps to extend the life of the filament in incandescent bulbs by reducing evaporation. In fluorescent lighting, argon is used to create a stable plasma that emits light.
- Scientific Research: Argon is used in various scientific experiments and equipment. For example, it is used in gas chromatography to separate and analyze different compounds.
- Food Packaging: Argon is used to displace oxygen in food packaging, helping to preserve the freshness and quality of the food by preventing oxidation and bacterial growth.
Argon's inert nature and stability make it an ideal choice for these applications. Its Argon # of Neutrons contributes to its overall stability and reliability, ensuring that it performs consistently in various environments.
Argon in the Atmosphere
Argon is the third most abundant gas in the Earth's atmosphere, making up about 0.934% of the air by volume. It is primarily produced through the decay of potassium-40 in the Earth's crust. This decay process releases argon-40 into the atmosphere, where it accumulates over time.
The presence of argon in the atmosphere is crucial for maintaining the Earth's climate and supporting life. Argon's inert nature means it does not react with other gases or compounds, making it a stable component of the atmosphere.
Argon's Argon # of Neutrons also plays a role in its atmospheric behavior. The stability of argon-40 ensures that it remains in the atmosphere for long periods, contributing to the overall composition of the air we breathe.
Isotopes of Argon
As mentioned earlier, argon has several isotopes, each with a different number of neutrons. The most common isotopes are argon-36, argon-38, and argon-40. Here is a table summarizing the key properties of these isotopes:
| Isotope | Number of Neutrons | Abundance | Stability |
|---|---|---|---|
| Argon-36 | 18 | 0.3365% | Stable |
| Argon-38 | 20 | 0.0632% | Stable |
| Argon-40 | 22 | 99.6003% | Stable |
While argon-36 and argon-38 are stable, they are present in much smaller quantities compared to argon-40. The stability of argon-40, with its 22 neutrons, makes it the dominant isotope in natural argon.
📝 Note: The abundance of argon isotopes can vary slightly depending on the source and measurement methods. However, argon-40 remains the most abundant isotope in all natural samples.
Argon in Geological Dating
Argon's isotopes, particularly argon-40, play a crucial role in geological dating. The decay of potassium-40 to argon-40 is used in potassium-argon (K-Ar) dating to determine the age of rocks and minerals. This method is based on the principle that the ratio of potassium-40 to argon-40 in a sample can be used to calculate the time since the sample solidified.
The Argon # of Neutrons in argon-40 is a key factor in this dating method. The stability of argon-40 ensures that it remains in the sample over time, providing an accurate measure of the decay process. This makes K-Ar dating a reliable method for determining the age of geological samples.
In addition to K-Ar dating, argon-argon (Ar-Ar) dating is another method that uses argon isotopes to determine the age of samples. This method involves irradiating the sample with neutrons to convert potassium-39 to argon-39, which can then be measured along with argon-40 to determine the age of the sample.
Both K-Ar and Ar-Ar dating methods rely on the stability and abundance of argon-40, making the Argon # of Neutrons a critical factor in these geological dating techniques.
📝 Note: Geological dating methods using argon isotopes require precise measurement and calibration to ensure accurate results. The stability of argon-40 is a key factor in the reliability of these methods.
Argon in Medical Applications
Argon's inert nature and stability make it useful in various medical applications. One of the most notable uses of argon in medicine is in argon plasma coagulation (APC). APC is a technique used to control bleeding during surgical procedures. It involves using a stream of argon gas to create a plasma that coagulates blood vessels, stopping the bleeding.
The Argon # of Neutrons in argon-40 contributes to its stability and inertness, making it safe for use in medical procedures. The stability of argon-40 ensures that it does not react with other compounds or tissues, reducing the risk of complications during surgery.
In addition to APC, argon is used in other medical applications, such as cryosurgery. Cryosurgery involves using extremely cold temperatures to destroy abnormal tissues, such as tumors. Argon is used to create a cold gas that is applied to the tissue, freezing and destroying the abnormal cells.
The stability and inertness of argon make it an ideal choice for these medical applications, ensuring that it performs consistently and safely.
📝 Note: The use of argon in medical applications requires careful control and monitoring to ensure safety and effectiveness. The stability of argon-40 is a key factor in its suitability for these applications.
Argon's unique properties, including its Argon # of Neutrons, make it a versatile and valuable element with a wide range of applications. From welding and lighting to geological dating and medical procedures, argon plays a crucial role in various industries and scientific fields. Its stability and inertness ensure that it performs consistently and reliably, making it an essential component in many technologies and processes.
Understanding the Argon # of Neutrons and its impact on the element’s properties provides insights into its behavior and applications. The stability of argon-40, with its 22 neutrons, is a key factor in its usefulness and reliability. Whether in industrial processes, scientific research, or medical procedures, argon’s unique characteristics make it an indispensable element in modern technology and science.
Related Terms:
- number of neutrons of argon
- amount of neutrons in argon
- argon protons and neutrons
- number of neutrons in argon
- argon protons electrons
- protons and neutrons in argon